TissueScope iQ Slide Scanner

Flexible scanning meets throughput and intelligence
Our newly-developed TissueScope iQ scanner combines flexibility and high throughput with smart features.
Download the TissueScope iQ Brochure here.
400 standard or 200 double-wide slides
TissueScope iQ features five slide cartridges, each holding 80 standard slides (400 total) or 40 double-wide slides (200 total). Standard or double-wide cartridges can be mixed and matched in the scanner for scanning flexibility.

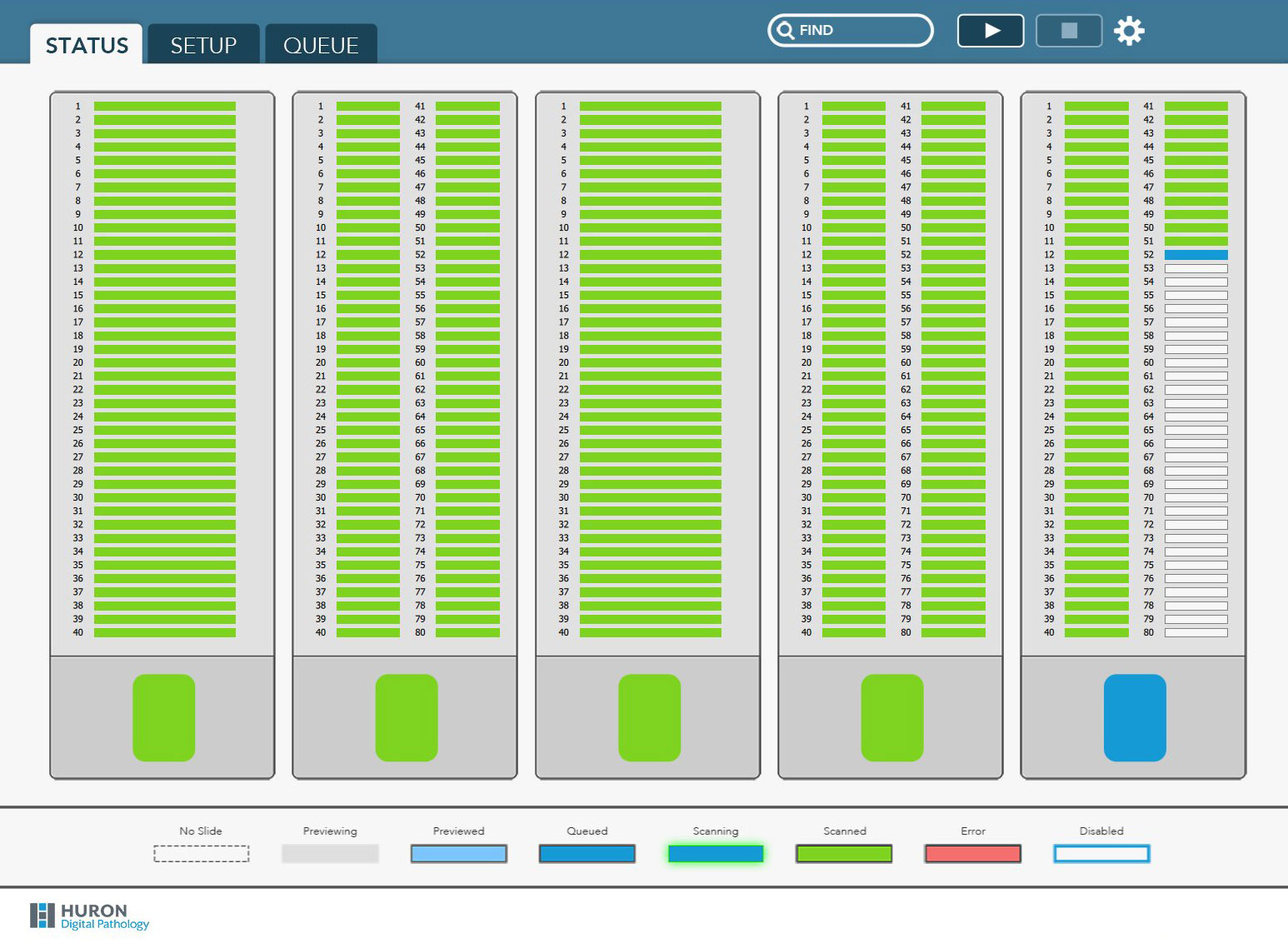
Fresh new user interface
Slide scanning has never been easier or more flexible. We completely refreshed the TissueScope iQ’s user interface so it is even more intuitive.
Choose “One-click” mode for fully automated scanning or expert mode for full scanning configurability.
TissueScope iQ Key Features
High capacity: up to 400 standard or 200 double-wide slides
Flexibility: Mix and match standard and double-wide
cartridge slidesNon-proprietary file format, DICOM compatible
Fresh, new user interface
Fast: <30 seconds per slide
Sharp, clear image quality up to 40X magnification
Z-Stack scanning
Image-based barcode reading
TissueScope iQ Technical Specifications
Imaging Mode
Brightfield
Slide Formats
25mm x 75mm (1″ x 3″)
50mm x 75mm (2″ x 3″)
Objective Resolution
20X 0.75NA
(40X 0.95NA Optional)
Resolution (µm/pixel)
0.25 at 40X
0.5 at 20X
Scan Speed
<30 seconds per slide, 15mm x 15mm @ 40X magnification
File Format
Non-proprietary 24-bit RGB Pyramidal BigTIFF
Uncompressed, JPEG or JPEG 2000 Compression
Export to JPEG, Flat TIFF and LZW Compressed TIFF
Dimensions & Weight
60cm (23.7″) Width
54cm (21.4″) Length
43 cm (16.9″) Height
44kg (97lbs) Weight
Warranty
One year
Please contact [email protected] for information on the use of the product in your facility. The regulatory status of the product differs by country/region.
The TissueScope™ scanner is based on one or more of the following: U.S. Patents 8,896,918 B2/ 8,655,043 B2/ 9,632,301/ 9,804,376 B2/ 9,575,304/ 10,114, 206/ 9,575,308/ 10,088,655/ 6,072,624;EU patents EP2758825 (A1)/ EP EP2656133 (A1)/ EP2864741 (A1); U.S. and International patents pending. Manufactured by Huron Technologies International Inc., of which Huron Digital Pathology is an operating division.